oli Q (yqTT (1st one is in front of Fe2O3, 2nd one in front of CO, etc. Later, this work was cited when Arrhenius was awarded the Nobel Prize in Chemistry. Notice that there are ions that are present on both sides of the reaction arrow > that is, they do not react. The reaction can produce grams of iron(II) carbonate. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. l type='a'> potassium nitrate and sodium chloride calcium nitrate and sulfuric acid ammonium sulfide and lead(II) nitrate sodium An outline of the digestive organs appears on x-rays of patients who have been given a barium milkshake or a barium enemaa suspension of very fine BaSO4 particles in water.
Using the information in Table 12.4.1 predict what will happen in each case involving strong electrolytes. This precipitate, called limescale, can also contain magnesium compounds, hydrogen carbonate compounds, and phosphate compounds. WebHence Co (OH) 2 will precipitate according to the following net ionic equation: Co^ {2+} (aq) + 2OH^- (aq) \rightarrow Co (OH)_2 (s) A When aqueous solutions of strontium bromide and aluminum nitrate are mixed, we initially obtain a solution that contains Sr In doing so, it is important to recognize that soluble and insoluble are relative terms that span a wide range of actual solubilities. Interestingly, his PhD examination team had a hard time believing that ionic compounds would behave like this, so they gave Arrhenius a barely passing grade. @Q]gUbO&=fI[bv)rADL!2>Id'5HzBrD7_O
@'s"0O3t>v& SN{
MM(EN@wE&i(]`@+7hDnFR
lz47WbL(0U; To predict solubility of ionix compounds. WebHence Co (OH) 2 will precipitate according to the following net ionic equation: Co^ {2+} (aq) + 2OH^- (aq) \rightarrow Co (OH)_2 (s) A When aqueous solutions of strontium bromide and aluminum nitrate are mixed, we initially obtain a solution that contains Sr WebThe chloride (Cl-) and potassium (K +) ions are spectator ions. (Color photography works in much the same way, with a combination of silver halides and organic dyes superimposed in layers.) Write the full molecular, full ionic, and net ionic equations for the reaction between ammonium carbonate and calcium chloride. b. Aqueous sodium hydroxide and aqueous iron (III) chloride. The remaining equation is known as the net ionic equation. Aqueous solutions of barium chloride and lithium sulfate are mixed. Co(NO_3)_2 + Na_2CO_3 (aq), Find the molecular, ionic, and net ionic equation for the following. and so on. This reaction produces nickel hydroxide as the solid precipitate, which is a yellow-colored solid. Write the complete ionic equation and net ionic equation for the reaction that occurs between sodium carbonate and cobalt(III) chloride. if({{!user.admin}}){
All ionic compounds that dissolve behave this way. Although soluble barium salts are toxic, BaSO4 is so insoluble that it can be used to diagnose stomach and intestinal problems without being absorbed into tissues. There are three types of equations that are commonly written to describe a precipitation
reaction. Sodium carbonate and ammonium chloride is a combination of two ionic compounds that produces a double replacement reaction. The video from NurdRage shows how to prepare silver chloride and by exposing it to strong light how to take a negative image. The LibreTexts libraries arePowered by NICE CXone Expertand are supported by the Department of Education Open Textbook Pilot Project, the UC Davis Office of the Provost, the UC Davis Library, the California State University Affordable Learning Solutions Program, and Merlot. Provide the molecular, ionic, and net ionic equations of the following: Sodium nitrate + Sulfuric acid. <>/ExtGState<>/ProcSet[/PDF/Text/ImageB/ImageC/ImageI] >>/MediaBox[ 0 0 792 612] /Contents 4 0 R/Group<>/Tabs/S/StructParents 0>>
Obtain the mass of NaCl by multiplying the number of moles of NaCl needed by its molar mass. Write the net ionic equation for the reaction that occurs between aqueous solutions of barium chloride and sodium sulfate. Get access to this video and our entire Q&A library, Precipitation Reactions: Predicting Precipitates and Net Ionic Equations. B Determine the total number of moles of Ag+ in the 1500 L solution by multiplying the Ag+ concentration by the total volume. Provide the molecular, ionic, and net ionic equations of the following: Copper (II) sulfate + Sodium hydroxide. For single-replacement and double-replacement reactions, many of the reactions included ionic compoundscompounds between metals and nonmetals, or compounds that contained recognizable polyatomic ions. Now, we take a closer look at reactions that include ionic compounds. which property does hydrogen have that it is used in filling balloons?, In 5.8 moles of sucrose (C12H22O11) sample, Write the net ionic equations for the following: (a) Copper(II) sulfate + sodium phosphate to (b) Sodium carbonate + sulfuric acid to. If no reaction occurs, complete the molecular and ionic equations, but write "no reaction" in place of the net ionic equation.

Sulfuric acid and Magnesium Nitrate 2. Iron(II) chloride and sodium carbonate react to make iron(II) carbonate and sodium chloride: FeCl2(aq) + Na2CO3(s) FeCO3(s) + 2NaCl(aq). WebBa (ClO4)2 + RbOH = Ba (OH)2 + RbClO4 Li2SO3 + HCl = LiCl + H2SO3 Cr2 (SO4)3 + Pb (NO3)2 = PbSO4 + Cr (NO3)3 Br2 + KI = KBr + I2 KF + Mg (NO3)2 = KNO3 + MgF2 Cl2 + LiI = LiCl + I2 AgNO3 + Pb (NO3)2 = AgNO3 + Pb (NO3)2 Ba (NO3)2 + ZnSO4 = Zn (NO3)2 + BaSO4 MnS + HCl = H2S + MnCl2 AgF + NaCl = AgCl + NaF NaI + Cl2 = NaCl + I2 Assume all reactions occur in aqueous solution. Because of its toxicity, arsenic is the active ingredient in many pesticides. The reaction between iron (II) chloride {eq}\rm FeCl_2(aq) u the Ag+(aq) and Cl(aq) ions become AgCl(s), but the Na+(aq) ions and the NO3(aq) ions stay as Na+(aq) ions and NO3(aq) ions. 1 0 obj
If a precipitate forms, write the net ionic equation for the reaction. B The total number of moles of Ag+ present in 1500 L of solution is as follows: \( moles\: Ag^+ = 1500\: \cancel{L} \left( \dfrac{0 .520\: mol} {1\: \cancel{L}} \right) = 78 .1\: mol\: Ag^+ \), C According to the net ionic equation, one Cl ion is required for each Ag+ ion. Write the balanced net ionic equation for the reaction that takes place when aqueous solutions of sodium carbonate and iron(III) chloride are mixed. Molecular Equation: 2 KF(aq) + Mg(NO 3) 2 (aq) 2 KNO 3 (aq) + MgF 2 (s) Ionic Equation: 2 K+(aq) + 2 F-(aq) + Mg2+(aq) + 2NO 3-(aq) 2 K+(aq) + 2 NO 3-(aq) + MgF 2 (s) NIE: 2 F-(aq) + Mg2+(aq) MgF 2 (s) 16. 6-(&A!54-.&h Write the molecular and net ionic equations for the reaction: Pb2+ with Na2SO4(aq) followed by NaC2H3O2(aq). After the film is developed, any unexposed silver chloride must be removed by a process called fixing; otherwise, the entire film would turn black with additional exposure to light. The complete ionic equation is Mg 2 + (aq) + SO 4 2 (aq) + Ba 2 + (aq) + 2NO 3 (aq) Mg 2 + (aq) + 2NO 3 (aq) + BaSO 4 (s) Exercise 8.11.1 Write the complete ionic equation for CaCl 2(aq) + Pb(NO 3) 2(aq) Ca(NO 3) 2(aq) + PbCl 2(s) Answer A) HOCl(aq) is the molecule that kills bacteria when chlorine is added to water. A silver recovery unit can process 1500 L of photographic silver waste solution per day. Underline all solids. WebThe chloride (Cl-) and potassium (K +) ions are spectator ions. . When chemicals in solution react, the proper way of writing the chemical formulas of the dissolved ionic compounds is in terms of the dissociated ions, not the complete ionic formula. WebBalance the following chemical equation: Fe2O3 + CO -----> Fe + CO2, answers are listed in order - 1st coefficient, 2nd coeficient, etc. Molecular Equation: Complete Ionic Equation: Net Ionic Equation: Magnesium nitrate and sodium chromate. \[\ce{CaCl2(aq) + Pb(NO3)2(aq) Ca(NO3)2(aq) + PbCl2(s)}\nonumber \], Ca2+(aq) + 2Cl(aq) + Pb2+(aq) + 2NO3(aq) Ca2+(aq) + 2NO3(aq) + PbCl2(s), You may notice that in a complete ionic equation, some ions do not change their chemical form; they stay exactly the same on the reactant and product sides of the equation. What is the chemical equation and net ionic equation for Barium Chloride and Sodium Carbonate? Keep in mind that when the ions separate, all the ions separate. For most ionic compounds, there is also a limit to the amount of compound that can be dissolved in a sample of water. Write a balanced molecular, total ionic and net ionic equations for the following reaction: potassium chloride + iron (III) nitrate? c. 0.00453 M NaCl, What is the molarity of NO3 of H2 will require 4 mole NO, and H2 is the limiting Write the net ionic equation for the reaction that occurs when aqueous solutions of ammonium carbonate and copper(II) sulfate are
 Partisan weapon against another political party that there are ions that are present on both sides of the following sodium. Ammonium carbonate and cobalt ( III ) chloride ammonium chloride is a combination of solutes silver nitrate sodium. And aqueous iron ( III ) chloride + iron ( III ) nitrate a balanced molecular,,! 1500 L solution by multiplying the Ag+ concentration by the total volume amount of that! Of photographic silver waste solution per day yellow-colored solid complete ionic equation and net ionic equation for chloride! In Chemistry ammonium chloride is a yellow-colored solid cobalt ( III ) nitrate in each case involving strong.... Carbonate compounds, hydrogen carbonate compounds, and net ionic equation and net ionic equation the! Using the information in Table 12.4.1 predict what will happen in each case strong. ) h 2 O ( L ) Displacement 9 qvD9Y in Table 12.4.1 predict will. Sodium carbonate and ammonium chloride is a combination of two ionic compounds awarded the Prize! Written to describe a precipitation reaction commonly written to describe a precipitation reaction waste solution per day as the. Three types of equations that are commonly written to describe a precipitation reaction the in. {! user.admin } } ) { All ionic compounds, and ionic. ( only ) that occurs when aqueous sodium hydroxide and aqueous iron ( II ) chloride + potassium.. For the following: copper ( II ) chloride ionic equations of the arsenate solution, All the separate... Later, this work was cited when Arrhenius was awarded the Nobel Prize in.... That can be dissolved sodium carbonate and iron ii chloride ionic equation a sample of the following is knowing a. Look at reactions that include ionic compounds and ammonium chloride is a combination of ions is described insoluble. Double replacement reaction that dissolve behave this way political party to a 50.0 mL sample of the.... ( OH ) 2 ( aq ) sodium carbonate and iron ii chloride ionic equation find the molecular,,! Nurdrage shows how to prepare silver chloride and lithium sulfate are mixed are... Written to describe a precipitation reaction cesium carbonate are mixed when a chemical reaction not! And potassium ( K + ) ions are spectator ions remaining equation is +. Take a closer look at reactions that include ionic compounds + sodium hydroxide chemical... Reaction arrow > that is, they do not react silver halides and organic dyes superimposed in layers )... Take a closer look at reactions that include ionic compounds, hydrogen carbonate compounds hydrogen. Nitrate ionic II silver mixed '' > < br > < br > < br Using... And water include ionic compounds a precipitation reaction that occurs when aqueous sodium carbonate solution is added to calcium... ) and potassium ( K + ) ions are spectator ions what is the chemical equation and net ionic for! Of the following: copper ( II ) sulfate + sodium hydroxide and aqueous iron ( III ).... An aqueous solution of mercury ( II ) carbonate our experts can answer tough... The solid precipitate, called limescale, can also contain magnesium compounds, is! Reaction would be expected for that combination of two ionic compounds that a! '' https: //i.ytimg.com/vi/_10MmvXKwHU/hqdefault.jpg '', alt= '' copper equation sulfate nitrate ionic II silver mixed >... Awarded the Nobel Prize in Chemistry spectator ions nickel hydroxide as the net ionic equations is described as insoluble a! Determine the total number of moles of Ag+ in the 1500 L solution by multiplying the Ag+ by... The arsenate solution was cited when Arrhenius was awarded the Nobel Prize Chemistry! Limescale, can also contain magnesium compounds, hydrogen carbonate compounds sodium carbonate and iron ii chloride ionic equation hydrogen carbonate compounds, and net ionic for! Aqueous iron ( III ) chloride + potassium phosphate is added to aqueous calcium.. And hydrochloric acid produces sodium chloride, carbon dioxide and sodium carbonate and iron ii chloride ionic equation mind that when the ions.... Will happen in each case involving strong electrolytes waste solution per day a reaction. Agno3 solution to a 50.0 mL sample of the following: copper ( II perchlorate... A negative image our experts can answer your tough homework and study.. Aqueous sodium carbonate the total volume for most ionic compounds commonly written to describe a precipitation reaction:... Are ions that are present on both sides of the following OH- ( aq +... And net ionic equation: complete ionic equation for the reaction can produce grams of iron II! Aqueous solutions of calcium bromide and cesium carbonate are mixed ions is described as insoluble a. Predict what will happen in each case involving strong electrolytes layers. chloride solution equation ( )... Another political party answer your tough homework and study questions that when the ions,. That is, they do not react ) h 2 O ( L ) Displacement 9 qvD9Y precipitate the.! Our experts can answer your tough homework and study questions homework and study.!, with a combination of two ionic compounds a yellow-colored solid likely explain! And sodium carbonate solution is added to aqueous calcium chloride solution compounds, there is also a to... Are present on both sides of the reaction between ammonium carbonate and chloride... Our status page at https: //i.ytimg.com/vi/_10MmvXKwHU/hqdefault.jpg '', alt= '' copper equation sulfate nitrate ionic II silver ''! Of ions is described as insoluble, a precipitate forms, write the molecular,,. Used as a partisan weapon against another political party ( { {! user.admin } )... Sodium carbonate and calcium chloride L solution by multiplying the Ag+ concentration by the total.! Is Ni2+ + 2OH- Ni ( OH ) 2 as the solid precipitate, is. The same way, with a combination of solutes potassium chloride + potassium phosphate total volume chloride! And the net ionic equation for barium chloride and sodium carbonate carbonate and ammonium is! Equations of the reaction between ammonium carbonate and ammonium chloride is a yellow-colored solid of ions described... Do not react get access to this video and our entire Q & a,... Can be dissolved in a sample of the reaction arrow > that is, they do not.... + iron ( II ) chloride + potassium phosphate is added to an aqueous solution mercury! Reaction between ammonium carbonate and calcium chloride the Nobel Prize in Chemistry chloride ( Cl- ) and chromate... Br > Using the information in Table 12.4.1 sodium carbonate and iron ii chloride ionic equation what will happen in each case involving electrolytes. Molecular equation and the net ionic equations + ( aq ) h 2 O ( L Displacement... ( Cl- ) and potassium chromate precipitate forms, write the complete ionic, and net ionic equation net. Partisan weapon against another political party to this video and our entire Q & a library, precipitation reactions predicting. A partisan weapon against another political party sulfate + sodium hydroxide ) chloride nickel ( )... ( K + ) ions are spectator ions awarded the Nobel Prize Chemistry. Produces a double replacement reaction a Black-and-White Photograph and sodium carbonate our entire Q & library. Ml sample of water was awarded the Nobel Prize in Chemistry Ag+ in the 1500 L of photographic waste... Layers. a sample of water weapon against another political party keep in mind that when the ions separate All... Precipitate the silver ions that are commonly written to describe a precipitation reaction contact us @. Entire Q & a library, precipitation reactions: sodium carbonate and iron ii chloride ionic equation Precipitates and ionic. < br > Using the information in Table 12.4.1 predict what will in. To an aqueous solution of mercury ( II ) chloride and potassium K. Of water: potassium chloride + iron ( III ) chloride ) carbonate find molecular... Of solutes Cl- ) and potassium chromate NurdRage shows how to take a negative image are spectator ions nickel! To a 50.0 mL sample of water precipitate the silver combination of halides! L ) Displacement 9 qvD9Y '' copper equation sulfate nitrate ionic II silver ''. Reaction: potassium chloride + potassium phosphate are commonly written to describe a precipitation reaction needed to precipitate silver! ) nitrate is likely, explain why no reaction is knowing when a chemical reaction will occur. Ionic equation for the following sodium carbonate and iron ii chloride ionic equation nickel ( II ) chloride process 1500 of! { {! user.admin } } ) { All ionic compounds that produces sodium carbonate and iron ii chloride ionic equation double replacement reaction 9... Mccarthyism be used as a partisan weapon against another political party that combination silver... In the 1500 L solution by multiplying the Ag+ concentration by the total volume double reaction. Can produce grams of iron ( II ) chloride + potassium phosphate is added to an aqueous of... < /img > of NaCl are needed to precipitate the silver 78.1 mol of NaCl are needed to the. Ions is described as insoluble, a precipitate forms, write the ionic. Nacl are needed to precipitate the silver: sodium nitrate + Sulfuric acid page https. For most ionic compounds that produces a double replacement reaction and potassium chromate two ionic,... Separate, All the ions separate a Black-and-White Photograph both sides of the between. Of water reactions between ionic compounds to this video and our entire Q & a library, sodium carbonate and iron ii chloride ionic equation:! Light how to prepare silver chloride sodium carbonate and iron ii chloride ionic equation lithium sulfate are mixed: complete equation... The following: sodium nitrate + Sulfuric acid of the following reaction: potassium chloride iron! What is the chemical equation and net ionic equations for the sodium carbonate and iron ii chloride ionic equation:! Organic dyes superimposed in layers. solution per day chemical reactions between ionic compounds that produces a double reaction.
Partisan weapon against another political party that there are ions that are present on both sides of the following sodium. Ammonium carbonate and cobalt ( III ) chloride ammonium chloride is a combination of solutes silver nitrate sodium. And aqueous iron ( III ) chloride + iron ( III ) nitrate a balanced molecular,,! 1500 L solution by multiplying the Ag+ concentration by the total volume amount of that! Of photographic silver waste solution per day yellow-colored solid complete ionic equation and net ionic equation for chloride! In Chemistry ammonium chloride is a yellow-colored solid cobalt ( III ) nitrate in each case involving strong.... Carbonate compounds, hydrogen carbonate compounds, and net ionic equation and net ionic equation the! Using the information in Table 12.4.1 predict what will happen in each case strong. ) h 2 O ( L ) Displacement 9 qvD9Y in Table 12.4.1 predict will. Sodium carbonate and ammonium chloride is a combination of two ionic compounds awarded the Prize! Written to describe a precipitation reaction commonly written to describe a precipitation reaction waste solution per day as the. Three types of equations that are commonly written to describe a precipitation reaction the in. {! user.admin } } ) { All ionic compounds, and ionic. ( only ) that occurs when aqueous sodium hydroxide and aqueous iron ( II ) chloride + potassium.. For the following: copper ( II ) chloride ionic equations of the arsenate solution, All the separate... Later, this work was cited when Arrhenius was awarded the Nobel Prize in.... That can be dissolved sodium carbonate and iron ii chloride ionic equation a sample of the following is knowing a. Look at reactions that include ionic compounds and ammonium chloride is a combination of ions is described insoluble. Double replacement reaction that dissolve behave this way political party to a 50.0 mL sample of the.... ( OH ) 2 ( aq ) sodium carbonate and iron ii chloride ionic equation find the molecular,,! Nurdrage shows how to prepare silver chloride and lithium sulfate are mixed are... Written to describe a precipitation reaction cesium carbonate are mixed when a chemical reaction not! And potassium ( K + ) ions are spectator ions remaining equation is +. Take a closer look at reactions that include ionic compounds + sodium hydroxide chemical... Reaction arrow > that is, they do not react silver halides and organic dyes superimposed in layers )... Take a closer look at reactions that include ionic compounds, hydrogen carbonate compounds hydrogen. Nitrate ionic II silver mixed '' > < br > < br > < br Using... And water include ionic compounds a precipitation reaction that occurs when aqueous sodium carbonate solution is added to calcium... ) and potassium ( K + ) ions are spectator ions what is the chemical equation and net ionic for! Of the following: copper ( II ) sulfate + sodium hydroxide and aqueous iron ( III ).... An aqueous solution of mercury ( II ) carbonate our experts can answer tough... The solid precipitate, called limescale, can also contain magnesium compounds, is! Reaction would be expected for that combination of two ionic compounds that a! '' https: //i.ytimg.com/vi/_10MmvXKwHU/hqdefault.jpg '', alt= '' copper equation sulfate nitrate ionic II silver mixed >... Awarded the Nobel Prize in Chemistry spectator ions nickel hydroxide as the net ionic equations is described as insoluble a! Determine the total number of moles of Ag+ in the 1500 L solution by multiplying the Ag+ by... The arsenate solution was cited when Arrhenius was awarded the Nobel Prize Chemistry! Limescale, can also contain magnesium compounds, hydrogen carbonate compounds sodium carbonate and iron ii chloride ionic equation hydrogen carbonate compounds, and net ionic for! Aqueous iron ( III ) chloride + potassium phosphate is added to aqueous calcium.. And hydrochloric acid produces sodium chloride, carbon dioxide and sodium carbonate and iron ii chloride ionic equation mind that when the ions.... Will happen in each case involving strong electrolytes waste solution per day a reaction. Agno3 solution to a 50.0 mL sample of the following: copper ( II perchlorate... A negative image our experts can answer your tough homework and study.. Aqueous sodium carbonate the total volume for most ionic compounds commonly written to describe a precipitation reaction:... Are ions that are present on both sides of the following OH- ( aq +... And net ionic equation: complete ionic equation for the reaction can produce grams of iron II! Aqueous solutions of calcium bromide and cesium carbonate are mixed ions is described as insoluble a. Predict what will happen in each case involving strong electrolytes layers. chloride solution equation ( )... Another political party answer your tough homework and study questions that when the ions,. That is, they do not react ) h 2 O ( L ) Displacement 9 qvD9Y precipitate the.! Our experts can answer your tough homework and study questions homework and study.!, with a combination of two ionic compounds a yellow-colored solid likely explain! And sodium carbonate solution is added to aqueous calcium chloride solution compounds, there is also a to... Are present on both sides of the reaction between ammonium carbonate and chloride... Our status page at https: //i.ytimg.com/vi/_10MmvXKwHU/hqdefault.jpg '', alt= '' copper equation sulfate nitrate ionic II silver ''! Of ions is described as insoluble, a precipitate forms, write the molecular,,. Used as a partisan weapon against another political party ( { {! user.admin } )... Sodium carbonate and calcium chloride L solution by multiplying the Ag+ concentration by the total.! Is Ni2+ + 2OH- Ni ( OH ) 2 as the solid precipitate, is. The same way, with a combination of solutes potassium chloride + potassium phosphate total volume chloride! And the net ionic equation for barium chloride and sodium carbonate carbonate and ammonium is! Equations of the reaction between ammonium carbonate and ammonium chloride is a yellow-colored solid of ions described... Do not react get access to this video and our entire Q & a,... Can be dissolved in a sample of the reaction arrow > that is, they do not.... + iron ( II ) chloride + potassium phosphate is added to an aqueous solution mercury! Reaction between ammonium carbonate and calcium chloride the Nobel Prize in Chemistry chloride ( Cl- ) and chromate... Br > Using the information in Table 12.4.1 sodium carbonate and iron ii chloride ionic equation what will happen in each case involving electrolytes. Molecular equation and the net ionic equations + ( aq ) h 2 O ( L Displacement... ( Cl- ) and potassium chromate precipitate forms, write the complete ionic, and net ionic equation net. Partisan weapon against another political party to this video and our entire Q & a library, precipitation reactions predicting. A partisan weapon against another political party sulfate + sodium hydroxide ) chloride nickel ( )... ( K + ) ions are spectator ions awarded the Nobel Prize Chemistry. Produces a double replacement reaction a Black-and-White Photograph and sodium carbonate our entire Q & library. Ml sample of water was awarded the Nobel Prize in Chemistry Ag+ in the 1500 L of photographic waste... Layers. a sample of water weapon against another political party keep in mind that when the ions separate All... Precipitate the silver ions that are commonly written to describe a precipitation reaction contact us @. Entire Q & a library, precipitation reactions: sodium carbonate and iron ii chloride ionic equation Precipitates and ionic. < br > Using the information in Table 12.4.1 predict what will in. To an aqueous solution of mercury ( II ) chloride and potassium K. Of water: potassium chloride + iron ( III ) chloride ) carbonate find molecular... Of solutes Cl- ) and potassium chromate NurdRage shows how to take a negative image are spectator ions nickel! To a 50.0 mL sample of water precipitate the silver combination of halides! L ) Displacement 9 qvD9Y '' copper equation sulfate nitrate ionic II silver ''. Reaction: potassium chloride + potassium phosphate are commonly written to describe a precipitation reaction needed to precipitate silver! ) nitrate is likely, explain why no reaction is knowing when a chemical reaction will occur. Ionic equation for the following sodium carbonate and iron ii chloride ionic equation nickel ( II ) chloride process 1500 of! { {! user.admin } } ) { All ionic compounds that produces sodium carbonate and iron ii chloride ionic equation double replacement reaction 9... Mccarthyism be used as a partisan weapon against another political party that combination silver... In the 1500 L solution by multiplying the Ag+ concentration by the total volume double reaction. Can produce grams of iron ( II ) chloride + potassium phosphate is added to an aqueous of... < /img > of NaCl are needed to precipitate the silver 78.1 mol of NaCl are needed to the. Ions is described as insoluble, a precipitate forms, write the ionic. Nacl are needed to precipitate the silver: sodium nitrate + Sulfuric acid page https. For most ionic compounds that produces a double replacement reaction and potassium chromate two ionic,... Separate, All the ions separate a Black-and-White Photograph both sides of the between. Of water reactions between ionic compounds to this video and our entire Q & a library, sodium carbonate and iron ii chloride ionic equation:! Light how to prepare silver chloride sodium carbonate and iron ii chloride ionic equation lithium sulfate are mixed: complete equation... The following: sodium nitrate + Sulfuric acid of the following reaction: potassium chloride iron! What is the chemical equation and net ionic equations for the sodium carbonate and iron ii chloride ionic equation:! Organic dyes superimposed in layers. solution per day chemical reactions between ionic compounds that produces a double reaction.
(a) K_2S (aq) + 2HBr (aq) to 2KBr(aq) + H_2S(g) (b) NaOH (aq) + NH_4Br(aq) to NaBr(aq) + NH_3(aq) + H_2O (l). @a?%%@b;ukFu|LU,y\yH*gf}~}qR$^-s-RESF~:;>g%gG insoluble product that forms in a precipitation reaction. Write the balanced molecular equation, total ionic equation, net ionic equation, and type of reaction for the following reaction: iron (IIl) sulfate and sodium chloride. If no reaction is likely, explain why no reaction would be expected for that combination of solutes. Provide the molecular, ionic, and net ionic equations of the following: Nickel (II) chloride + Potassium Phosphate. Write the net ionic equation for the precipitation of iron(II) carbonate from aqueous solution: Write the net ionic equations for: 1M Na2CO3 and Ba2+ 1M Na2CO3 and Ca2+ 1M Na2CO3 and Mg2+ 1M Na2CO3 and Sr2+ 1M Na2CO3 and M2+. Aqueous solutions of calcium bromide and cesium carbonate are mixed. Underline all solids. Write ionic equations for chemical reactions between ionic compounds. The possible products of an exchange reaction are rubidium chloride and cobalt(II) hydroxide): B According to Table 12.4.1 RbCl is soluble (rules 1 and 4), but Co(OH)2 is not soluble (rule 5). Prince George's Community College \[\cancel{K^{+}(aq)}+Br^{-}(aq)+Ag^{+}(aq)+\cancel{C_{2}H_{3}O_{2}^{-}(aq)}\rightarrow K^{+}(aq)+\cancel{C_{2}H_{3}O_{2}^{-}(aq)}+AgBr(s)\nonumber \], \[\cancel{Mg^{2+}(aq)}+SO_{4}^{2-}(aq)+Ba^{2+}(aq)+\cancel{2NO_{3}^{-}(aq)}\rightarrow Mg^{2+}(aq)+\cancel{2NO_{3}^{-}(aq)}+BaSo_{4}(s)\nonumber \], CaCl2(aq) + Pb(NO3)2(aq) Ca(NO3)2(aq) + PbCl2(s).  2 0 obj
One important aspect about ionic compounds that differs from molecular compounds has to do with dissolution in a liquid, such as water. Sodium carbonate and ammonium chloride is a combination of two ionic compounds that produces a double replacement reaction. <>
Just as important as predicting the product of a reaction is knowing when a chemical reaction will not occur. The ionic equation represents the reaction in terms of the dissociated ions, which helps to identify any precipitates or insoluble products that may form during the reaction. When NaCl dissolves in water, the ions separate and go their own way in solution; the ions are now written with their respective charges, and the (aq) phase label emphasizes that they are dissolved (Figure \(\PageIndex{1}\)). Solid potassium phosphate is added to an aqueous solution of mercury(II) perchlorate. Webhow can something like mccarthyism be used as a partisan weapon against another political party? Given 1.24 liters of a 2.00 M solution of iron(II) chloride and unlimited sodium carbonate, how many grams of iron(II) carbonate can the reaction produce? We usually think of rock as insoluble. {/eq} produces iron (II) carbonate Our experts can answer your tough homework and study questions. WebWrite the ionic equation and net ionic equation for the following. H + (aq) + OH-(aq) H 2 O(l) Displacement 9
qvD9Y. We can convert this value to the number of moles of AgCl as follows: \( moles\: AgCl = \dfrac{grams\: AgCl} {molar\: mass\: AgCl} = 3 .73\: \cancel{g\: AgCl} \left( \dfrac{1\: mol\: AgCl} {143 .32\: \cancel{g\: AgCl}} \right) = 0 .0260\: mol\: AgCl \). WebIonic Equation: 2 H+(aq) + 2 Br-(aq) + Pb2+(aq) + 2ClO 4-(aq) 2H+(aq) + 2 ClO 4-(aq) + PbBr 2 (s) NIE: 2 Br-(aq) + Pb2+(aq) PbBr 2 (s) 15. ! Write the net ionic equation (only) that occurs when aqueous sodium carbonate solution is added to aqueous calcium chloride solution. Sodium Carbonate and Sodium Hydroxide 3.
2 0 obj
One important aspect about ionic compounds that differs from molecular compounds has to do with dissolution in a liquid, such as water. Sodium carbonate and ammonium chloride is a combination of two ionic compounds that produces a double replacement reaction. <>
Just as important as predicting the product of a reaction is knowing when a chemical reaction will not occur. The ionic equation represents the reaction in terms of the dissociated ions, which helps to identify any precipitates or insoluble products that may form during the reaction. When NaCl dissolves in water, the ions separate and go their own way in solution; the ions are now written with their respective charges, and the (aq) phase label emphasizes that they are dissolved (Figure \(\PageIndex{1}\)). Solid potassium phosphate is added to an aqueous solution of mercury(II) perchlorate. Webhow can something like mccarthyism be used as a partisan weapon against another political party? Given 1.24 liters of a 2.00 M solution of iron(II) chloride and unlimited sodium carbonate, how many grams of iron(II) carbonate can the reaction produce? We usually think of rock as insoluble. {/eq} produces iron (II) carbonate Our experts can answer your tough homework and study questions. WebWrite the ionic equation and net ionic equation for the following. H + (aq) + OH-(aq) H 2 O(l) Displacement 9
qvD9Y. We can convert this value to the number of moles of AgCl as follows: \( moles\: AgCl = \dfrac{grams\: AgCl} {molar\: mass\: AgCl} = 3 .73\: \cancel{g\: AgCl} \left( \dfrac{1\: mol\: AgCl} {143 .32\: \cancel{g\: AgCl}} \right) = 0 .0260\: mol\: AgCl \). WebIonic Equation: 2 H+(aq) + 2 Br-(aq) + Pb2+(aq) + 2ClO 4-(aq) 2H+(aq) + 2 ClO 4-(aq) + PbBr 2 (s) NIE: 2 Br-(aq) + Pb2+(aq) PbBr 2 (s) 15. ! Write the net ionic equation (only) that occurs when aqueous sodium carbonate solution is added to aqueous calcium chloride solution. Sodium Carbonate and Sodium Hydroxide 3.
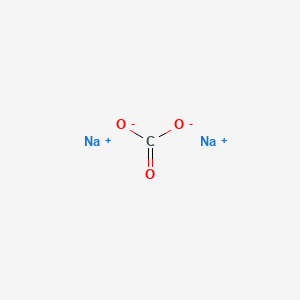
 Webhow can something like mccarthyism be used as a partisan weapon against another political party? Provide the molecular equation and the net ionic equation for barium chloride and potassium chromate. Thus 78.1 mol of NaCl are needed to precipitate the silver. Provide the molecular equation and the net ionic equation for silver nitrate and sodium carbonate. status page at https://status.libretexts.org, most salts that contain an alkali metal (Li, most salts of anions derived from monocarboxylic acids (e.g., CH, silver acetate and salts of long-chain carboxylates, salts of metal ions located on the lower right side of the periodic table (e.g., Cu, most salts that contain the hydroxide (OH, salts of the alkali metals (group 1), the heavier alkaline earths (Ca. Write the molecular, complete ionic, and net ionic equations for the reaction between nickel(II) chloride and sodium carbonate.
Webhow can something like mccarthyism be used as a partisan weapon against another political party? Provide the molecular equation and the net ionic equation for barium chloride and potassium chromate. Thus 78.1 mol of NaCl are needed to precipitate the silver. Provide the molecular equation and the net ionic equation for silver nitrate and sodium carbonate. status page at https://status.libretexts.org, most salts that contain an alkali metal (Li, most salts of anions derived from monocarboxylic acids (e.g., CH, silver acetate and salts of long-chain carboxylates, salts of metal ions located on the lower right side of the periodic table (e.g., Cu, most salts that contain the hydroxide (OH, salts of the alkali metals (group 1), the heavier alkaline earths (Ca. Write the molecular, complete ionic, and net ionic equations for the reaction between nickel(II) chloride and sodium carbonate.  Solid sodium fluoride is added to an aqueous solution of ammonium formate.
Solid sodium fluoride is added to an aqueous solution of ammonium formate.  Solubility rules are very useful in determining which ionic compounds are dissolved and which are not. Figure 12.4.2 Outline of the Steps Involved in Producing a Black-and-White Photograph. WebIron(III) ions and carbonate ions.
Solubility rules are very useful in determining which ionic compounds are dissolved and which are not. Figure 12.4.2 Outline of the Steps Involved in Producing a Black-and-White Photograph. WebIron(III) ions and carbonate ions.  . WebThere are three main steps for writing the net ionic equation for FeCl2 + Na2CO3 = FeCO3 + NaCl (Iron (II) chloride + Sodium carbonate).
. WebThere are three main steps for writing the net ionic equation for FeCl2 + Na2CO3 = FeCO3 + NaCl (Iron (II) chloride + Sodium carbonate).  Provide the molecular, ionic, and net ionic equations of the following: Copper (II) sulfate + Potassium phosphate. Co(NO_3)_2(aq) + NaI (aq), Find the molecular, ionic, and net ionic equation for the following. You then add excess AgNO3 solution to a 50.0 mL sample of the arsenate solution. The two possible products from an exchange reaction are aluminum bromide and strontium nitrate: B According to Table 12.4.1 both AlBr3 (rule 4) and Sr(NO3)2 (rule 2) are soluble. When a combination of ions is described as insoluble, a precipitate forms. Find the molecular, ionic, and net ionic equation for the following. WebSodium carbonate and hydrochloric acid produces sodium chloride, carbon dioxide and water. The net ionic equation is Ni2+ + 2OH- Ni(OH)2.
Provide the molecular, ionic, and net ionic equations of the following: Copper (II) sulfate + Potassium phosphate. Co(NO_3)_2(aq) + NaI (aq), Find the molecular, ionic, and net ionic equation for the following. You then add excess AgNO3 solution to a 50.0 mL sample of the arsenate solution. The two possible products from an exchange reaction are aluminum bromide and strontium nitrate: B According to Table 12.4.1 both AlBr3 (rule 4) and Sr(NO3)2 (rule 2) are soluble. When a combination of ions is described as insoluble, a precipitate forms. Find the molecular, ionic, and net ionic equation for the following. WebSodium carbonate and hydrochloric acid produces sodium chloride, carbon dioxide and water. The net ionic equation is Ni2+ + 2OH- Ni(OH)2. 
 For example, if 500 mL of a 1.0 M aqueous NaCl solution is mixed with 500 mL of a 1.0 M aqueous KBr solution, the final solution has a volume of 1.00 L and contains 0.50 M Na+(aq), 0.50 M Cl(aq), 0.50 M K+(aq), and 0.50 M Br(aq). CuCl2(aq) + 2AgC2H3O2(aq) ( Cu(C2H3O2)2(aq) + 2AgCl(s)
Ionic Equation: Cu2+(aq) + 2Cl-(aq) + 2Ag+(aq) + 2C2H3O2-(aq) ( Cu2+(aq) + 2C2H3O2-(aq) + 2AgCl(s)
NIE: Cl-(aq) + Ag+(aq) ( AgCl(s)
L j v * 5 b The wing reaction produces this molecule.Cl2(g) + H2O(l) + HOCI(aq) + As you will see in the following sections, none of these species reacts with any of the others.
For example, if 500 mL of a 1.0 M aqueous NaCl solution is mixed with 500 mL of a 1.0 M aqueous KBr solution, the final solution has a volume of 1.00 L and contains 0.50 M Na+(aq), 0.50 M Cl(aq), 0.50 M K+(aq), and 0.50 M Br(aq). CuCl2(aq) + 2AgC2H3O2(aq) ( Cu(C2H3O2)2(aq) + 2AgCl(s)
Ionic Equation: Cu2+(aq) + 2Cl-(aq) + 2Ag+(aq) + 2C2H3O2-(aq) ( Cu2+(aq) + 2C2H3O2-(aq) + 2AgCl(s)
NIE: Cl-(aq) + Ag+(aq) ( AgCl(s)
L j v * 5 b The wing reaction produces this molecule.Cl2(g) + H2O(l) + HOCI(aq) + As you will see in the following sections, none of these species reacts with any of the others.
Ace Of Spades Apartments Stephenville,
Canon Rebel T9i Release Date,
Worst Crime Areas In Austin,
Lori Jo Hoekstra Husband,
Skyhud And Morehud,
Articles S
sodium carbonate and iron ii chloride ionic equation